WasteLog®
Centralized Assay & Verification for Clear Injectable Medications
WasteLog® supports pharmacy teams with instant verification of wasted and returned clear injectable medications

Overview
WasteLog® is a centralized pharmacy assay solution designed to verify wasted, returned or prepared clear injectable medications as part of a comprehensive QC and diversion control strategy.
Operated by pharmacy staff or pharmacy technicians, WasteLog® enables accurate verification of:
- Assay device for returned and wasted controlled substance syringes
- QC device used for compounded preparations
- Clear injectable medications prepared outside the perioperative area
Unlike the Safe Waste System™ (SWS) — which is designed for anesthesiologist-operated identification, measurement, waste, assay and reconciliation of clear liquid controlled substances at the point of care immediately following a case — WasteLog® is implemented in the pharmacy or satellite pharmacy for centralized verification and analysis.
Designed for the Pharmacy Environment
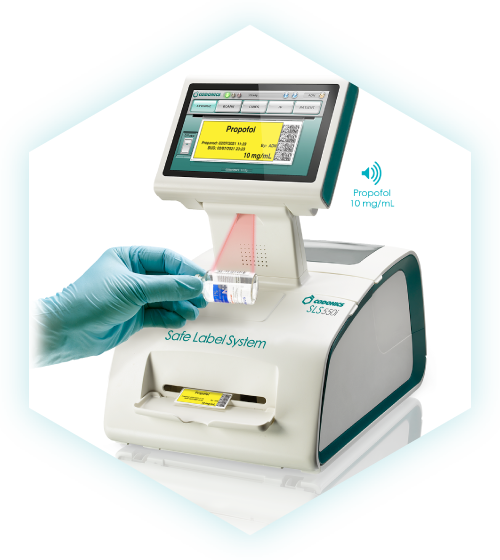
WasteLog® uses UV-visible absorption spectroscopy to analyze a small sample (0.3–0.5 mL) drawn from a syringe.
- Assay results delivered in <4 seconds
- Accurate identification of low-dose concentrations
- Clear pass/fail determination
- Immediate spectral curve visualization
- Data exportable to third-party analytics systems
- The cuvette-based design supports instrument cleanliness and sample retention when further review is required.
New drug calibrations can be created onsite, allowing pharmacy teams to maintain control as formularies evolve.
Request A Demo
How it works
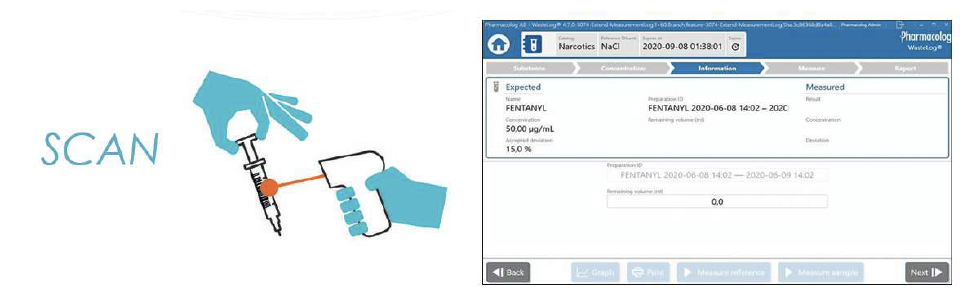
Once In Pharmacy, or anywhere returns occur, wasted syringes are scanned, enabling instant electronic identification of the drug name and concentration of the syringe contents.
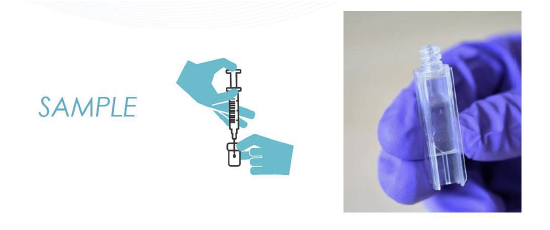
A small sample (0.3-0.5mL) is injected into the cuvette.

WasteLog® uses UV and visible light absorption spectroscopy to analyze the sample and measure concentrations. It is seen as a stronger technique than Raman or Refractometry, two other types of technologies to determine substances.

With the cuvette placed in the WasteLog unit, results are provided in seconds. The cuvettes keep the machine clean and reduce contamination and can be capped and saved for further analysis.
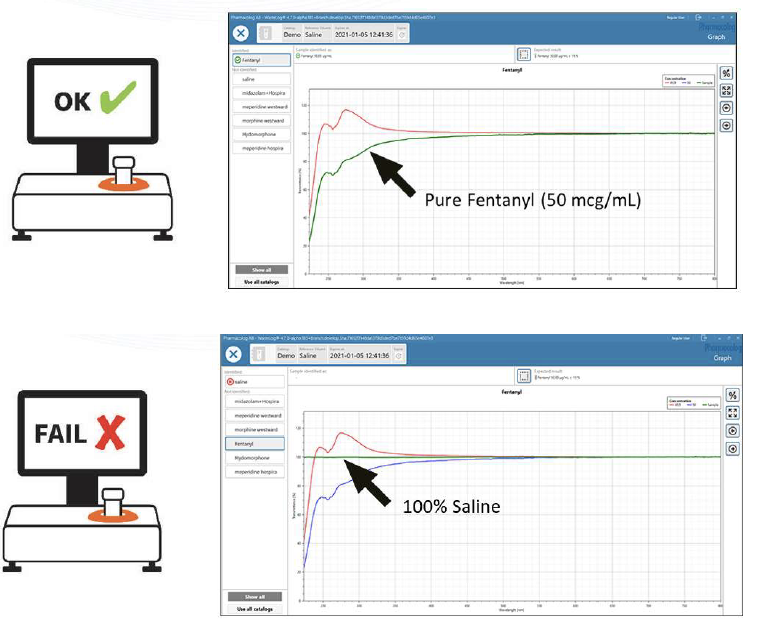
Samples either pass or fail. A few clicks on the touch screen initiates the test. A failed result gives indicators and provides opportunity for in-depth analysis. Immediate viewing the curves can help to understand the failed result.
Platform Integration
When used alongside Codonics Safe Label System® (SLS), pharmacy staff can scan the machine-readable SLS prepared syringe label to electronically capture the drug name and concentration into WasteLog®, helping streamline the assay workflow and improve documentation accuracy.
Hospitals may implement WasteLog® independently or as part of a broader Codonics medication safety strategy to provide a robust, proven and modern approach to drug diversion.

WasteLog® and Safe Waste System™ (SWS)
Distinct Workflows. Complementary Roles.
Hospitals may choose one solution — or both — based on workflow goals.
| WasteLog® | Safe Waste System™ (SWS) | |
|---|---|---|
| Centralized pharmacy verification | Point-of-care controlled substance waste & reconciliation | |
| Technology | UV-Vis Spectrophotometer | UV-Vis Spectrophotometer |
| Primary User | Pharmacy / Pharmacy Tech | Anesthesia Provider |
| Location | Centralized Pharmacy / Satellite Pharmacy | Perioperative environment / Point-of-care |
| Timing | Post-return / Centralized verification | Immediately after the case |
| Medication Type | Any clear injectable medication | Clear liquid-controlled substances |
| Function | Assay & verify | Identify, measure, waste, assay & reconcile at the point-of-care |
Supporting a Structured Diversion Control Approach
Some hospitals implement Safe Waste System™ (SWS) in the perioperative environment to manage liquid controlled substance waste, assay, and reconciliation in real time.
Others implement WasteLog® within pharmacy to assay returned syringes, QC compounded medications or clear injectables prepared outside the perioperative area.
Many hospitals implement both — establishing point-of-care waste workflows while maintaining centralized pharmacy assay capabilities.
Together, these technologies support a coordinated, accountable medication handling strategy across departments.
Product Videos
WasteLog Support and Resources
Patient Safety Products
Designed for Clinicians, Driven by Pharmacy.
Improving healthcare from drug preparation to administration and waste.